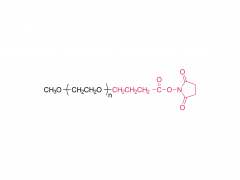
I. Product Overview
This series of products represents a class of elegantly designed and functionally powerful heterobifunctional crosslinkers. Each molecule features an Fmoc (fluorenylmethoxycarbonyl) protecting group and an NHS (N-hydroxysuccinimide ester) active ester at its two termini, connected by PEG (polyethylene glycol) spacers of varying lengths. This unique design makes them indispensable tools in fields such as chemical biology, drug delivery, materials science, and cutting-edge PROTAC technology.
II. Structural Features
Dual Functionality: Enables two sequential, specific reactions.
Excellent Water Solubility: The PEG chain significantly improves the overall water solubility of the molecule, enhancing reaction efficiency.
Cleavable Linker: The Fmoc group can be rapidly and completely removed under mild basic conditions (e.g., piperidine/DMF), revealing a free amino group for controlled cleavage or functional switching.
High Flexibility: Available in different PEG lengths to optimize steric hindrance, hydrophilicity, and molecular size.
III. Major Application Areas
Chemical Biology & Protein Modification
Protein/Antibody Labeling and PEGylation: Introduce PEG chains and potential subsequent functional groups onto proteins via the NHS ester to improve pharmacokinetics, increase stability, or introduce reaction handles.
Peptide Synthesis and Modification: Used as a deprotectable linking unit or to introduce functional PEG spacers in solid‑phase or solution‑phase peptide synthesis.
Materials Science & Bioconjugation
Functionalization of Material Surfaces: Couple the NHS ester to amine‑modified nanoparticles, chips, hydrogels, etc., then remove Fmoc to generate free amino groups on the surface for further immobilization of ligands, drugs, or fluorescent dyes.
Controlled Assembly Systems: Exploit the two‑step reaction property to construct sequentially assembled or “unlockable” supramolecular structures.
Drug Delivery Systems
Prodrugs and Linkers: Act as cleavable linkers to conjugate drug molecules (via the exposed amino group) to targeting moieties or carriers (via the NHS ester). The PEG chain enhances water solubility, and Fmoc removal can trigger drug release or activation.
IV. Applications in PROTAC Technology
PROTAC (Proteolysis‑Targeting Chimera) is a revolutionary targeted protein degradation technology. Fmoc‑PEGn‑NHS serves as a cleavable, length‑tunable linker in this field.
“Click Chemistry” Strategy: Commonly used to synthesize E3 ligase ligands or target protein ligands containing an amino group. First, the amino group of a ligand reacts with the NHS ester of Fmoc‑PEGn‑NHS to form a stable, easily purified, and storable Fmoc‑protected PEGylated ligand intermediate.
Modular Assembly: When needed, the Fmoc group is removed to afford a PEGylated ligand bearing a free amino group. This amino group can then be conveniently condensed with another ligand module containing a carboxyl group (or an activated ester) to efficiently and modularly assemble the complete PROTAC molecule.
Linker Length Optimization: PEG chain length is a critical factor influencing the formation efficiency of the ternary PROTAC complex (event‑driven pharmacodynamics) and cell permeability. The Fmoc‑PEGn‑NHS series enables systematic screening of different PEG lengths to identify the optimal linker for inducing target protein degradation.
Improved Physicochemical Properties: Introduction of the PEG chain enhances the water solubility and drug‑like properties of PROTAC molecules, especially for highly hydrophobic combinations.